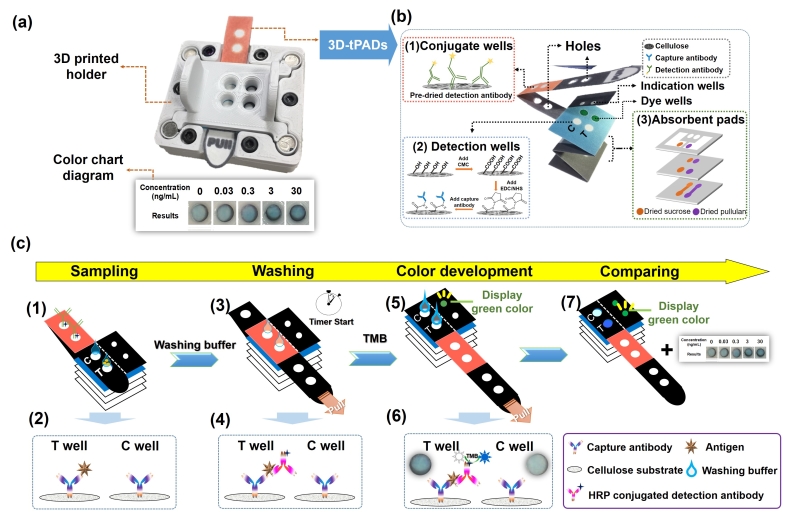
本研究受摺紙藝術啟發,透過簡單的紙張折疊與滑動之設計以及比色顯示,並於其中整合了一不需要電力之「計時器」以指示免疫反應中各步驟之執行時間,研發出可攜式傳染病免疫檢測試紙,可直接透過肉眼觀察半定量出血液中之致病菌含量。此項與傳染科盛望徽老師合作之成果於2021年5月刊登英國皇家化學會Lab on a chip期刊,並獲選為該期封底以及收錄於年度論文集 (Lab on a Chip HOT Articles 2021)。
致病菌是能夠引起人類疾病之微生物,其中包括細菌、病毒以及真菌等。人體一旦受到感染,病原體會透過不斷增殖及變異,增強其致病性與傳染力。為遏止致病菌的傳播,相關的精準即時檢測平臺顯得尤為重要。本研究團隊開發一種快速、便宜、可攜式、低樣品使用劑量、且無需專業儀器輔助之致病菌檢測試紙元件,能達到無需運送樣品至實驗室即可現場進行檢測,適合於醫療匱乏地區對當地患者進行準確即時的診斷與自身病情監控,避免延誤治療造成擴大傳染之情況產生。
本平臺由層析紙張構成,偵測區域進行化學表面修飾,因而可以特異性地吸附免疫分析所需之專一抗體試劑,減少紙張檢測過程中非特異性吸附問題,可提升檢測之靈敏度與專一性。此外,藉由改變液體於紙張元件中流動體積,以及乾燥蔗糖與普魯蘭多糖等於流動過程中以控制檢測試劑於多層紙張內流動之流速,達到於不同時間潤濕染劑顯色而準確告知測試者其免疫反應所需操作以及用肉眼讀取判讀顏色變化結果的時間,並以3D列印製造夾具來穩定流體於紙張元件內之流動性質,免除了使用計時器以及高價量測設備的需要。研究以HIV表面蛋白p24為設定之檢測目標,成功於十分鐘內可完成一無需任何儀器之免疫檢測流程,將來並可簡單透過改變免疫測試中的抗原抗體組合,使其進一步可應用於其他傳染病於各種資源有限環境下之免疫快速篩檢。
本研究感謝科技部,以及臺灣大學高教深耕計劃之經費支持。
執行單位
工學院 | 應力所 | 陳建甫